
If you’re looking for more help with IB Chemistry, including hints for your IA, then be sure to visit our Chemistry subject page where you’ll find more articles, study tools, and other resources. A table of contents for the book as well as some sample pages are available through the link below. If you’re looking for practice questions on this topic or more succinct explanations of key concepts in IB Chemistry Option C, then pick up a copy of our study guide. This quick review of nuclear fusion and fission has hopefully helped to reinforce your understanding. Expanding your IB Chemistry Option C: Energy knowledge You do not need to learn any specific nuclear equations but you do need to be able to deduce mass numbers and atomic numbers or the name of the elements for the nuclei involved, if given enough information. In your exam you might be set problems ‘ balancing’ these nuclear equations. Nuclear fusion occurs when two very light nuclei (such as hydrogen) combine together to make a larger nucleus.Įnormous heat and pressure are required to overcome the electrical repulsion of protons but there is a net release of energy in the process. There are two ways this can happen: nuclear fusion and nuclear fission. Nuclear energy is energy that is released when atomic nuclei change. The guide also contains over 25 practice questions plus four exam-style questions to allow you to test your knowledge and understanding. The study guide is full of concise, easy-to-understand explanations of complicated concepts that can help you stay on top of your IB Chemistry Option C studies. If you’re studying IB Chemistry Option C: Energy then this straightforward explanation of nuclear fusion and nuclear fission extracted from our IB study guide, Chemistry Option C: Energy Standard and Higher Level written by Tony Hickling, is for you. File:Nuclear fission.IB Chemistry is known for being a challenging subject, so we’ve pulled some useful explanations of key concepts from our study guides to help make your revision a bit easier.License: CC BY-SA: Attribution-ShareAlike License: CC BY-SA: Attribution-ShareAlikeĬC LICENSED CONTENT, SPECIFIC ATTRIBUTION nucleon: One of the subatomic particles of the atomic nucleus (i.e., a proton or a neutron).fission: The process of splitting the nucleus of an atom into smaller particles nuclear fission.fusion: A nuclear reaction in which nuclei combine to form more massive nuclei with the concomitant release of energy. 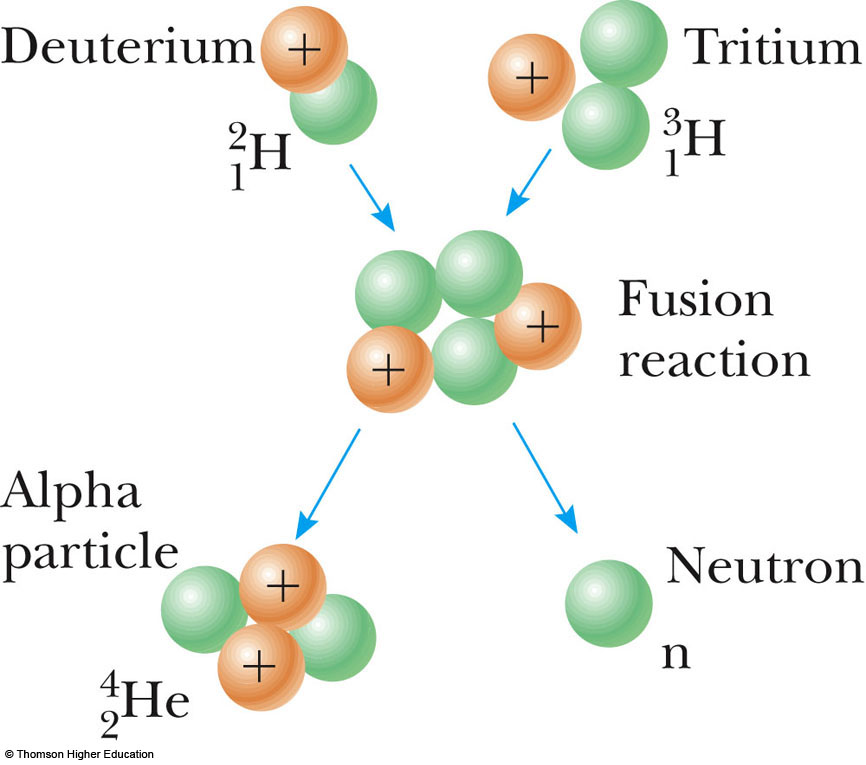
tunneling: the quantum-mechanical passing of a particle through an energy barrier.Nuclear fusion and fission involve the conversion of matter to energy, but the matter that is converted is never a full nucleon.In radioactive decay, a proton can be converted to a neutron and a neutron can be converted to a proton (beta-decay).Splitting of 92 U 235 by absorbing neutron is best example of nuclear fission reaction. There is also release of 200MeV energy per fission. The law of Conservation of Nuclear Number states that the sum of protons and neutrons among species before and after a nuclear reaction will be the same. Nuclear fission reaction of Uranium-235 When 92 U 235 is bombarded with thermal neutron, it splits up into 56 Ba 141 and 36 Kr 92 with the emission of 3 neutrons. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
